Eli Lilly antibody drug 'significantly' reduces risk of COVID-19 in nursing homes, study shows
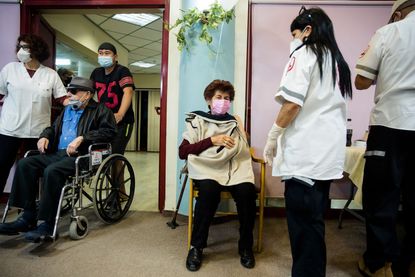

Eli Lilly's antibody drug bamlanivimab was found to "significantly" reduce the risk of contracting symptomatic COVID-19 in nursing homes, the company has announced.
Eli Lilly said Thursday a phase 3 trial showed that nursing home residents who preventively received bamlanivimab rather than a placebo had an up to 80 percent lower risk of contracting symptomatic COVID-19. Additionally, the company said bamlanivimab reduced the risk by 57 percent for both residents and staff, The Wall Street Journal reports.
"We are exceptionally pleased with these positive results, which showed bamlanivimab was able to help prevent COVID-19, substantially reducing symptomatic disease among nursing home residents, some of the most vulnerable members of our society," Eli Lilly chief scientific officer Daniel Skovronsky said.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
Skovronsky further explained in an interview with the Journal that the company's antibody treatment is "not an alternative for a vaccine," but "for people who haven't been vaccinated, and there's an outbreak in their facility — this could be a last resort." In Eli Lilly's announcement, Skovronsky said that after the Food and Drug Administration previously provided an emergency use authorization for bamlanivimab, the company hopes to work "with regulators to explore expanding the emergency use authorization to prevent the spread of COVID-19 in these facilities."
Create an account with the same email registered to your subscription to unlock access.
Sign up for Today's Best Articles in your inbox
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Brendan worked as a culture writer at The Week from 2018 to 2023, covering the entertainment industry, including film reviews, television recaps, awards season, the box office, major movie franchises and Hollywood gossip. He has written about film and television for outlets including Bloody Disgusting, Showbiz Cheat Sheet, Heavy and The Celebrity Cafe.
-
 Justices set to punt on Trump immunity case
Justices set to punt on Trump immunity caseSpeed Read Conservative justices signaled support for Trump's protection from criminal charges
By Peter Weber, The Week US Published
-
 Do youth curfews work?
Do youth curfews work?Today's big question Banning unaccompanied children from towns and cities is popular with some voters but is contentious politically
By Harriet Marsden, The Week UK Published
-
 'Sleaze baack!'
'Sleaze baack!'Today's Newspapers A roundup of the headlines from the US front pages
By The Week Staff Published
-
 Texas dairy worker gets bird flu from infected cow
Texas dairy worker gets bird flu from infected cowSpeed Read The virus has been spreading among cattle in Texas, Kansas, Michigan and New Mexico
By Peter Weber, The Week US Published
-
 Dengue hits the Americas hard and early
Dengue hits the Americas hard and earlySpeed Read Puerto Rico has declared an epidemic as dengue cases surge
By Peter Weber, The Week US Published
-
 US bans final type of asbestos
US bans final type of asbestosSpeed Read Exposure to asbestos causes about 40,000 deaths in the U.S. each year
By Peter Weber, The Week US Published
-
 Seattle Children's Hospital sues Texas over 'sham' demand for transgender medical records
Seattle Children's Hospital sues Texas over 'sham' demand for transgender medical recordsSpeed Read Texas Attorney General Ken Paxton subpoenaed records of any Texan who received gender-affirming care at the Washington hospital
By Peter Weber, The Week US Published
-
 Afghanistan has a growing female suicide problem
Afghanistan has a growing female suicide problemSpeed Read The Taliban has steadily whittled away women's and girls' rights in Afghanistan over the past 2 years, prompting a surge in depression and suicide
By Peter Weber, The Week US Published
-
 US life expectancy rose in 2022 but not to pre-pandemic levels
US life expectancy rose in 2022 but not to pre-pandemic levelsSpeed Read Life expectancy is slowly crawling back up
By Devika Rao, The Week US Published
-
 Vallance diaries: Boris Johnson 'bamboozled' by Covid science
Vallance diaries: Boris Johnson 'bamboozled' by Covid scienceSpeed Read Then PM struggled to get his head around key terms and stats, chief scientific advisor claims
By The Week UK Published
-
 An increasing number of dog owners are 'vaccine hesitant' about rabies
An increasing number of dog owners are 'vaccine hesitant' about rabiesSpeed Read A new survey points to canine vaccine hesitancy
By Devika Rao, The Week US Published