Moderna to start clinical trials of booster shot that targets COVID-19 variant

Moderna announced on Wednesday it will begin testing a new version of its COVID-19 vaccine designed to target the coronavirus variant first reported in South Africa.
The pharmaceutical company said it has sent doses of the booster shot to the U.S. National Institutes of Health for clinical trials. In a statement, CEO Stéphane Bancel said Moderna is "committed to making as many updates to our vaccine as necessary until the pandemic is under control."
Moderna previously revealed that preliminary studies showed the vaccine still made neutralizing antibodies above protective levels for the South African variant, but because it was a reduced level, it prompted the company to begin tweaking the vaccine against the strain, as well as variants that first spread in the United Kingdom and Brazil.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
Moderna is already conducting tests involving giving a third dose of its original vaccine as a booster to people who have received two doses, and the new clinical trials will evaluate the safety of the variant-specific booster and a "multivalent booster candidate" that is one dose of the variant-specific booster and original vaccine, NBC News reports.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Catherine Garcia has worked as a senior writer at The Week since 2014. Her writing and reporting have appeared in Entertainment Weekly, The New York Times, Wirecutter, NBC News and "The Book of Jezebel," among others. She's a graduate of the University of Redlands and the Columbia University Graduate School of Journalism.
-
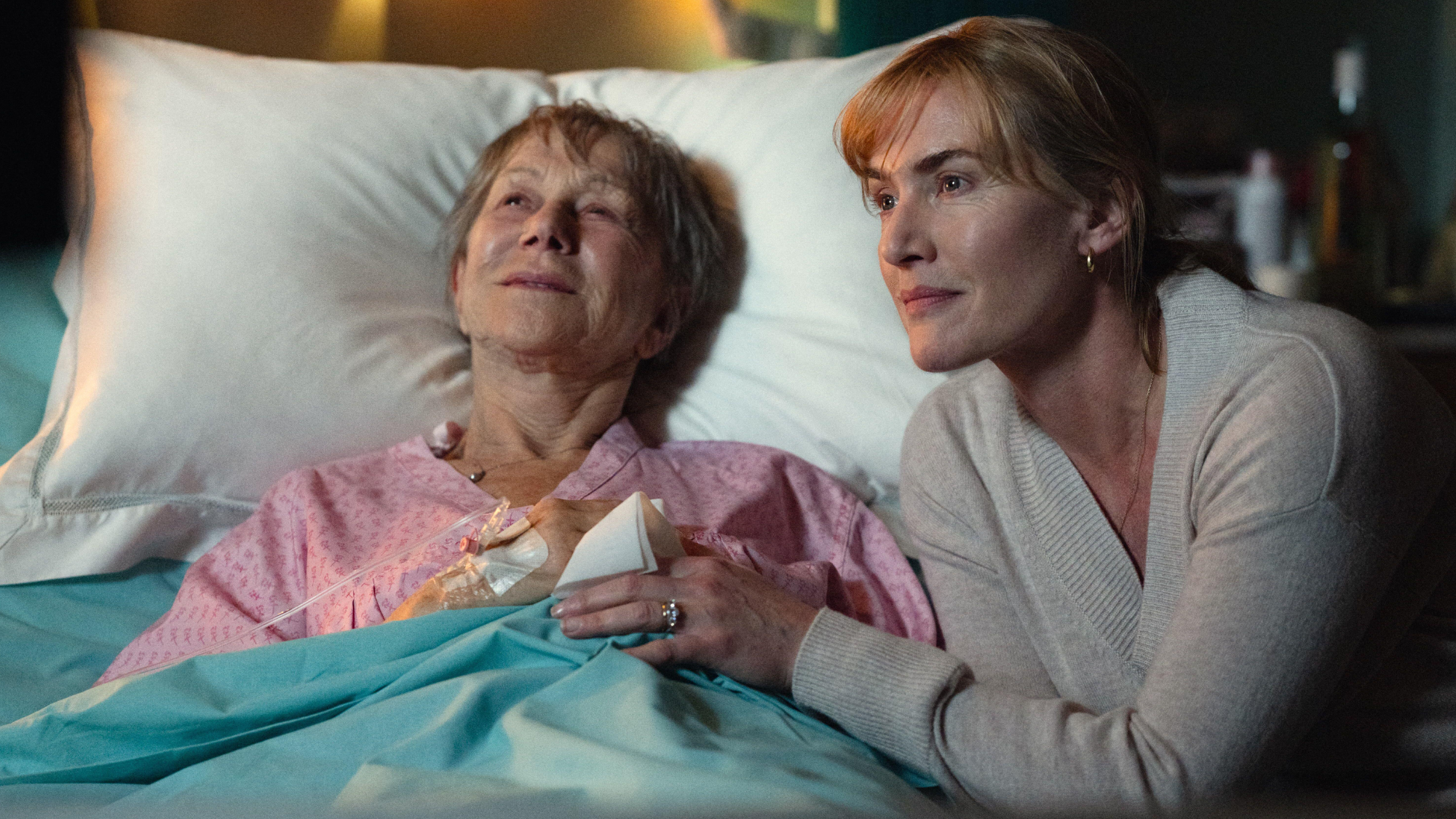 Goodbye June: Kate Winslet’s directorial debut feels like a ‘John Lewis Christmas TV ad’
Goodbye June: Kate Winslet’s directorial debut feels like a ‘John Lewis Christmas TV ad’Talking Point Helen Mirren stars as the terminally ill English matriarch in this sentimental festive heartwarmer
-
 A Christmas Carol (or two)
A Christmas Carol (or two)The Week Recommends These are the most delightful retellings of the Dickens classic from around the country
-
 Political cartoons for December 18
Political cartoons for December 18Cartoons Thursday’s political cartoons include Susie Wiles under scrutiny, Venezuela's oil, and more
-
 FDA OKs generic abortion pill, riling the right
FDA OKs generic abortion pill, riling the rightSpeed Read The drug in question is a generic version of mifepristone, used to carry out two-thirds of US abortions
-
 RFK Jr. vaccine panel advises restricting MMRV shot
RFK Jr. vaccine panel advises restricting MMRV shotSpeed Read The committee voted to restrict access to a childhood vaccine against chickenpox
-
 Texas declares end to measles outbreak
Texas declares end to measles outbreakSpeed Read The vaccine-preventable disease is still spreading in neighboring states, Mexico and Canada
-
 RFK Jr. shuts down mRNA vaccine funding at agency
RFK Jr. shuts down mRNA vaccine funding at agencySpeed Read The decision canceled or modified 22 projects, primarily for work on vaccines and therapeutics for respiratory viruses
-
 Measles cases surge to 33-year high
Measles cases surge to 33-year highSpeed Read The infection was declared eliminated from the US in 2000 but has seen a resurgence amid vaccine hesitancy
-
 Kennedy's vaccine panel signals skepticism, change
Kennedy's vaccine panel signals skepticism, changeSpeed Read RFK Jr.'s new vaccine advisory board intends to make changes to the decades-old US immunization system
-
 Kennedy ousts entire CDC vaccine advisory panel
Kennedy ousts entire CDC vaccine advisory panelspeed read Health Secretary RFK Jr. is a longtime anti-vaccine activist who has criticized the panel of experts
-
 RFK Jr. scraps Covid shots for pregnant women, kids
RFK Jr. scraps Covid shots for pregnant women, kidsSpeed Read The Health Secretary announced a policy change without informing CDC officials
