Will Contrave revolutionize dieting?
The FDA may approve the first new weight-loss drug in more than a decade — raising hopes that a pill could solve America's obesity problem
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
You are now subscribed
Your newsletter sign-up was successful
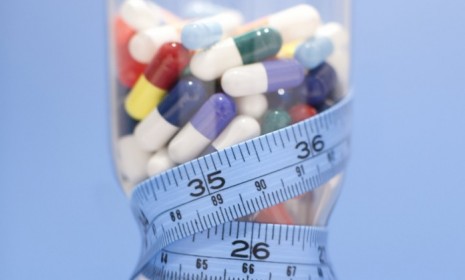
America is in the throes of an obesity "epidemic," but the famously wary FDA hasn't approved a new diet pill in more than a decade. That may change with Orexigen Therapeutics' debut drug, Contrave, which an FDA panel recommended for approval Tuesday. The FDA itself still has to sign off on the drug, but tends to follow the panel's recommendations. How does Contrave work, and will it be the silver bullet for America's weight problem? (Watch a Fox Business discussion about skepticism over Contrave)
What's Contrave's secret?
It combines two existing drugs: Bupropion (aka Wellbutrin), the popular antidepressant and smoking-cessation medication, and naltrexone, used to counter alcohol addiction. This combination apparently boosts metabolism while decreasing appetite to help people lose weight.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
Are the side effects bad?
Since Contrave contains two long-established drugs, the panel decided the risks are pretty well understood, and clinical trials suggest that the negative effects of combining the two are pretty modest: A slight increase in blood pressure and pulse, which in theory could contribute to heart attacks or strokes. Bupropion can also cause seizures. The FDA panel, while positive, did recommend that Orexigen conduct longer, wider studies after Contrave is approved.
How effective is it?
Slightly better than the traditional diet strategy: Eating healthier and exercising more. The trial participants who received Contrave knocked an average of 4.2 percentage points more off their weight than those taking placebos. That's less than the FDA likes, but the pill passed muster because at least 35 percent of Contrave-takers lost 5 percent or more of their weight during the year-long trial. "I think [Orexigen] made it by the hair of their chinny chin chin," says Melanie G. Coffin, the patient representative on the panel. "It's sad to see that that is as far as they could go."
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
What other diet pills are out there?
The only prescription diet pill on the U.S. market is the rarely prescribed Xenical, which can cause liver problems, gas, and uncontrolled bowel movements. The entire U.S. weight-loss drug market is only about $382 million annually, according to IMS Health. If Contrave is approved (and is tolerated as well as expected), it could reach sales of $1.2 billion by 2018, forecasts BioMedTracker.
Given the obesity crisis, why are there so few pills?
The FDA has typically deemed diet pills too risky. Merida, one that was approved (in 1997), was just pulled from the shelves in October after studies showed it increased the risk of strokes and heart attacks. Anti-obesity groups say the FDA has been too cautious, or set the bar too high. "This panel has voted against every obesity treatment that has come before it this year," anti-obesity advocate Theodore K. Kyle told the panel. "I ask you to take the disease seriously and take the benefits of treatment seriously."
Will Contrave revolutionize weight-loss?
Not necessarily. It will be one of the "very few options" available to those for whom weight loss is "not just a matter of willpower," says Kate James in Gather.com. "The main advice is diet and exercise, which is great and can help. However, there are obviously some people who need additional help." Financially, Contrave will undoubtedly "be wildly successful, at least at first," says University of Florida pharmacy professor Paul Doering. Will it work as a diet pill? Time will tell, but "I'm going to let somebody else be the first to take it."
Sources: New York Times, Reuters/MSNBC, ABC News, Gather
-
 Can Europe regain its digital sovereignty?
Can Europe regain its digital sovereignty?Today’s Big Question EU is trying to reduce reliance on US Big Tech and cloud computing in face of hostile Donald Trump, but lack of comparable alternatives remains a worry
-
 The Mandelson files: Labour Svengali’s parting gift to Starmer
The Mandelson files: Labour Svengali’s parting gift to StarmerThe Explainer Texts and emails about Mandelson’s appointment as US ambassador could fuel biggest political scandal ‘for a generation’
-
 Magazine printables - February 13, 2026
Magazine printables - February 13, 2026Puzzle and Quizzes Magazine printables - February 13, 2026