How do nuclear power plants work?
The answer is simpler than you might imagine


Nuclear reactors undergo a surprisingly simple process to generate power. In fact, it's quite similar to the process for coal-driven plants: Water is heated to make steam, and the steam drives a turbine to produce electricity. The key difference is how the heat is created. In a nuclear power plant, the heat to make steam is produced through nuclear fission, when an atom is split into two and releases energy.
The atoms being split in today's nuclear plants are those of the metal uranium. Natural uranium, which is relatively abundant, is a composite of two isotopes: It's 99.3 percent uranium-238 and .7 percent uranium-235. "A nuclear reactor can't operate with natural uranium," explains Dr. David Brenner, Director of the Center for Radiological Research and a radiation biophysics professor at Columbia University. "It has to have a bigger proportion of uranium-235 to operate…uranium-235 is the relevant nuclear fuel." The uranium-235 must be boosted up from its natural .7 percent concentration to about 3 percent, through a process called enrichment. (Weapons-grade uranium has to be enriched further still, which is why there is so much international concern about which powers have enrichment capabilities, like Iran.)
It's worth noting that uranium undergoes spontaneous fission all the time, and emits radiation in the process, but it does so at a very slow rate. When nuclear power plants induce fission to create heat and energy, they are simply speeding up the process through a "chain reaction" process. The enriched uranium is pressed into fuel pellets and placed into thin rods, roughly "the diameter of a pencil and about couple of meters long," as Dr. Brenner described.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
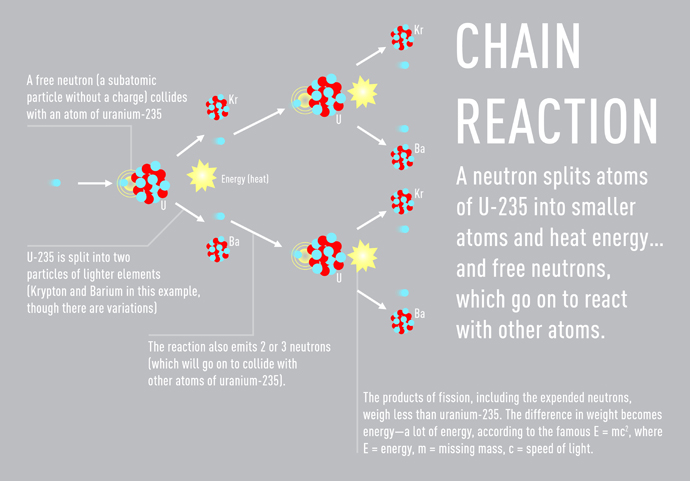
The rods are coated in zirconium, a metal that absorbs relatively few of the neutrons produced in a fission reaction and is highly resistant to heat and chemical erosion. An assembly of about 200 of these rods becomes what's called the "nuclear core" of the reactor, and atom-splitting neutrons are fired into the core. When an atom of uranium-235 is split (into one Barium and one Krypton atom), the reaction releases energy, heat, and neutrons that then are free to collide with other uranium-235 atoms. A chain reaction begins, and — when carefully controlled, as with interfering "control rods" that can be inserted into the core to slow down a potentially runaway chain reaction — the result is exploitable nuclear energy.
More from World Science Festival...
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
