Coming soon: An over-the-counter HIV test?
The FDA takes a step toward approving the first at-home kit to detect the virus. How important will this be in the battle against AIDS?
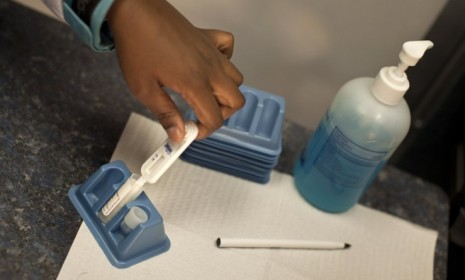
In a potentially huge step in the fight against AIDS, the advisory committee for the Food and Drug Administration has unanimously voted to recommend approval for the first over-the-counter HIV test. Carl Schmid, deputy director of the AIDS Institute, said the kit — the OraQuick In-Home HIV Test — could be a "game-changer." Could this really prove effective in preventing the transmission of AIDS? Here, a concise guide to this potential breakthrough:
How does the test work?
It's simple. If the kit gets final approval, you'll be able to pick one up at the local drug store. To take the test, the user takes a mouth swab, collecting fluid to be checked for the HIV virus. The result is ready in 20 minutes.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
Is it reliable?
A version of the test marketed to doctors, which sells for $17.50, has proven 99 percent accurate. The at-home version, which is expected to sell for under $60, delivered a correct result about 93 percent of the time.
But aren't false results potentially deadly?
Yes, says Cassie Murdoch at Jezebel. After all, a false negative result "might lead someone who actually is infected to potentially expose other people without knowing it." The FDA estimates that roughly 50,000 people would take the test in a year, meaning around 3,800 might feel reassured that they are HIV-negative, when in reality they're infected.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Then why approve the tests?
One-fifth of the 1.2 million Americans who are HIV-positive — about 240,000 people — don't know they're infected. Those people are responsible for up to 70 percent of the 50,000 new infections each year. The FDA estimates that if 50,000 people take the test in a year, 4,000 new infections will be prevented, because more HIV-positive people will know to seek treatment and take precautions to protect themselves and others. "Having a home test kit for HIV," Dr. Arthur Caplan of the University of Pennsylvania says at MSNBC, "is a bit like relying on a bathroom scale in the battle against obesity."

