Scotland approves breast cancer drug deemed 'too expensive' for England
Kadcyla to be offered north of the border, while NHS Scotland also commits to funding 'costly' PrEP HIV drug
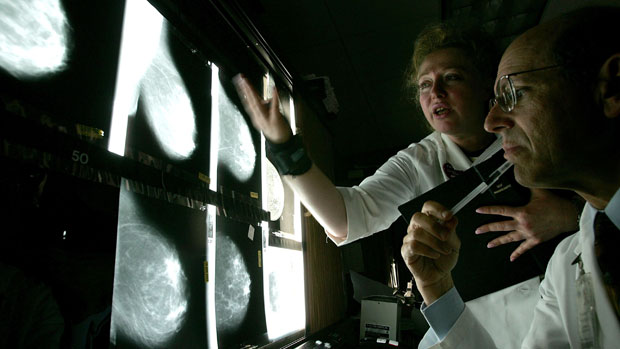
Scotland's health service has approved a breast cancer drug that is being withdrawn in England for being too expensive.
Kadcyla has been shown to extend the lives of patients dying from an aggressive form of breast cancer by an average of nine months.
The Scottish Medicines Consortium (SMC) said the drug would allow patients "more time with their families and in some cases return to work".
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
However, last year the National Institute for Health and Care Excellence (Nice), which decides which drugs and treatments are available on the NHS, said Kadcyla was too expensive.
More than 13,000 women have signed a petition calling for the drug to be made routinely available. A decision on whether to approve its availability will be made this summer, said Nice.
NHS Scotland has also become the first health service in the UK to fund the use of a drug that reduces the risk of HIV infection by 90 per cent. Pre-exposure prophylaxis (PrEP) will be free to those who need it within weeks.
The SMC's ruling follows "a fierce legal battle south of the border last year", says the Daily Telegraph, when NHS England unsuccessfully argued it was not responsible for providing the medicine.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Health officials south of the border have resisted approving PrEP due to its cost, "although it is a lot cheaper than a lifetime of HIV treatment which could cost £360,000", The Guardian reports.
PrEP has not yet been approved for routine preventative use, although following the fight last year, NHS England said it would make the drug available through a trial involving at least 10,000 people over the next three years.
Aids campaigners believe the SMC decision will put pressure on England's health service to change the way it deals with preventative HIV measures.
Deborah Gold, chief executive of the National Aids Trust, said: "This game-changing prevention tool has the potential to massively reduce HIV rates and turn Scotland into a model internationally of how to do HIV prevention well.
"The speed and decisiveness of the Scottish process contrasts starkly with delays in the other three UK nations."
