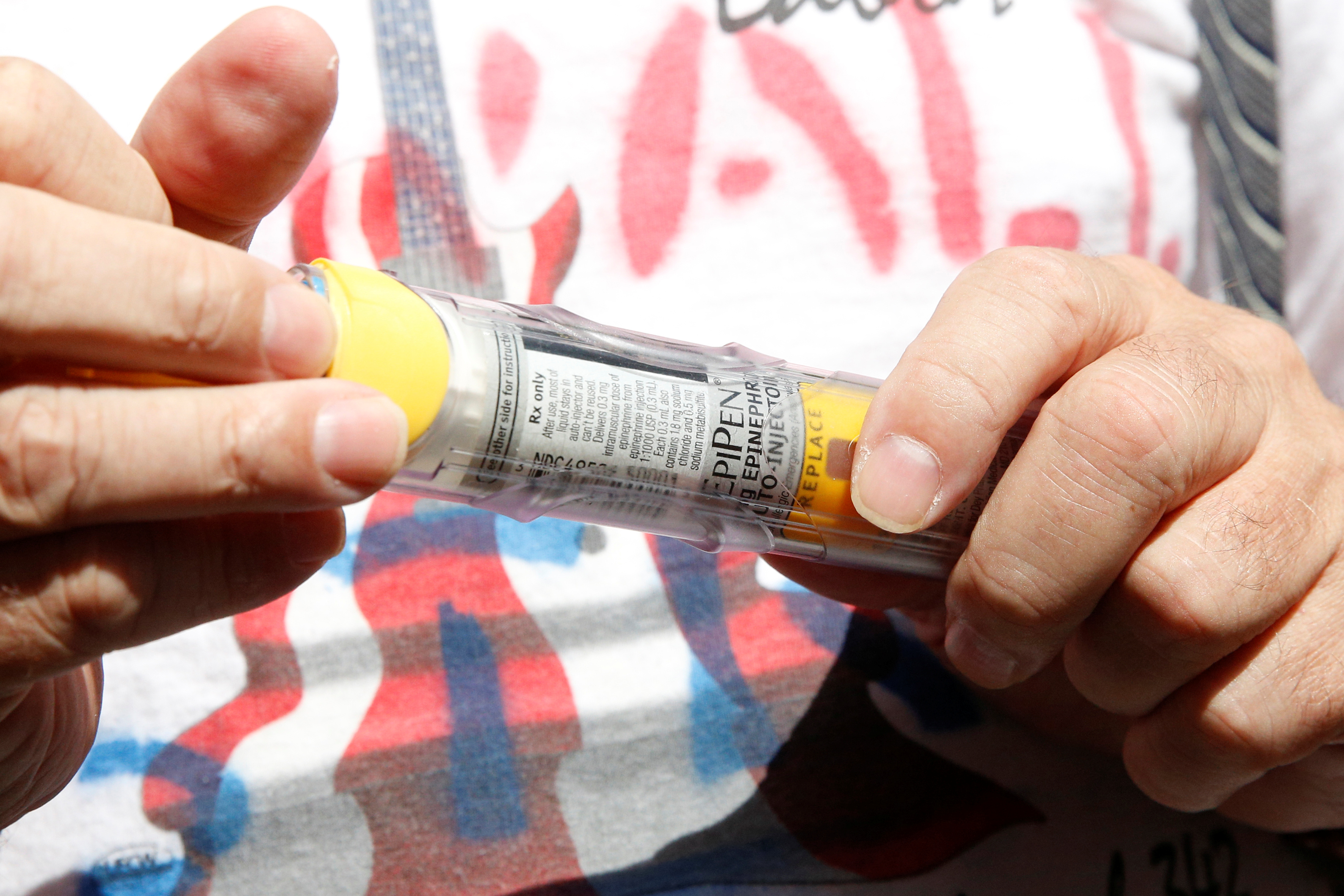
The EpiPen uproar, explained
The EpiPen now costs about $600 for a two-pack — up from roughly $100 in 2007
The smartest insight and analysis, from all perspectives, rounded up from around the web:
Move over, Martin Shkreli, America has a new drug industry villain, said Jordan Weissmann at Slate. Mylan Pharmaceuticals has drawn "public wrath and congressional scrutiny" over the relentless series of price hikes on its lifesaving EpiPen. The device, which stops potentially fatal allergic reactions by injecting a precise dose of epinephrine, now costs about $600 for a two-pack, up from roughly $100 when Mylan acquired EpiPen in 2007. Mylan CEO Heather Bresch — who earned nearly $19 million in total compensation last year — initially tried to defend the company, blaming its price rises on a "broken" health-care system. But after weeks "of thorough public shaming, it looks like Mylan is kind of, sort of, throwing in the towel." The company announced this week that it would soon release a generic version of the EpiPen, which will cost only $300 per two-pack. So, progress?
"EpiPens are a perfect example of a health-care nightmare," said Aaron Carroll at The New York Times. Epinephrine itself is very cheap, costing less than $1 per milliliter, and there's less than a third of that in an EpiPen. But to save the life of someone suffering a severe allergic reaction to a bee sting, drugs, or food, epinephrine must be injected quickly and in the right dose. That's why the proprietary EpiPen auto-injector is needed. Mylan is guaranteed to sell plenty of units because epinephrine degrades quickly, so users have to replace their EpiPens every year. Kids also need them at home, at school, at camp, and at Grandma's, so people often buy quite a few at once. Because the EpiPen has virtually no competitors and health insurers often cover a big chunk of its cost, Mylan has been able to "raise the price again and again with little pushback."
Don't expect a generic EpiPen to change things, said Dante Ramos at The Boston Globe. Mylan's cheaper offering is intended to get Congress off the company's back, and is "a shrewd business move" from a firm that lowered its tax bill by moving its corporate address from the U.S. to the Netherlands in 2015. The $300 price tag is high enough to generate plenty of profit — it's more than a brand-name EpiPen cost only a few years ago — but low enough to deter other firms from taking on the risk of developing their own generic. Meanwhile, some consumers' irrational preference for brand-name drugs means that Mylan can still expect healthy sales of its expensive EpiPen. "'How can we even think of cheaper substitutes,' an anxious parent might reason, 'when our child's life might be at stake?'"
"Rather than focusing on the price tag, we should be asking why EpiPens face no real market competition," said Paul Howard at The Hill. It's not for lack of effort. Auvi-Q, an auto-injector made by France's Sanofi, was recalled after patients reported receiving inaccurate doses. Adrenaclick, another competitor, isn't considered "therapeutically equivalent" to the EpiPen, so doctors tend not to prescribe it. But the biggest obstacle is the regulation imposed by the U.S. Food and Drug Administration. One company estimates that it would cost at least $1.5 billion to develop an EpiPen alternative and push it through clinical trials. "Instead of pointing fingers, we need to push for reforms that allow new competitors to enter the market more quickly."
Editor's note: This article originally misstated the potential cost of developing an EpiPen alternative. It has since been corrected. We regret the error.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.