FDA backs Pfizer COVID booster for 12- to 15-year-olds

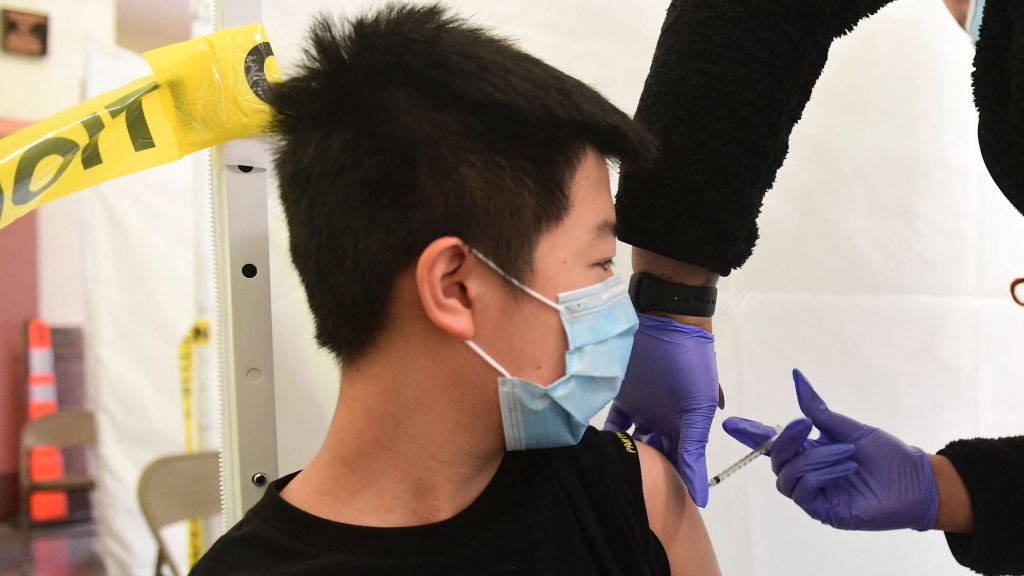
The U.S. Food and Drug Administration on Monday officially authorized Pfizer-BioNTech COVID-19 boosters for use in children ages 12 to 15, The Washington Post reports, "an effort to bolster protection as schools reopen amid a surge of infections caused by the Omicron variant."
According to CNBC, the FDA also cut down the recommended time between primary vaccination and an individual's booster dose from six months to at least five, and authorized a "third vaccine dose as part of the primary series of shots for children ages 5 through 11 who have compromised immune systems."
The agency's verdict arrives as schools decide whether and how to reopen schools while an Omicron surge tears its way through the country, per the Post. According to the Centers for Disease Control and Prevention, about one-third of eligible Americans have thus far received a COVID booster shot.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
"The recent rise in COVID-19 cases is concerning to all and today's decision by the FDA to further expand the Emergency Use Authorization of a booster dose of our vaccine is critical to help us ultimately defeat this pandemic," said Pfizer CEO Albert Bourla in a statement.
The expansion will now be reviewed by the CDC and its panel of vaccine advisers likely later this week, notes the Post. Should the outside committee sign off on the additional shots, "CDC director Rochelle Walensky is expected to officially recommend them" that same day.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Brigid Kennedy worked at The Week from 2021 to 2023 as a staff writer, junior editor and then story editor, with an interest in U.S. politics, the economy and the music industry.

