World's first malaria vaccine Mosquirix to be tested in Africa
Ghana, Kenya and Malawi to pilot new medication which could help save thousands of lives each year
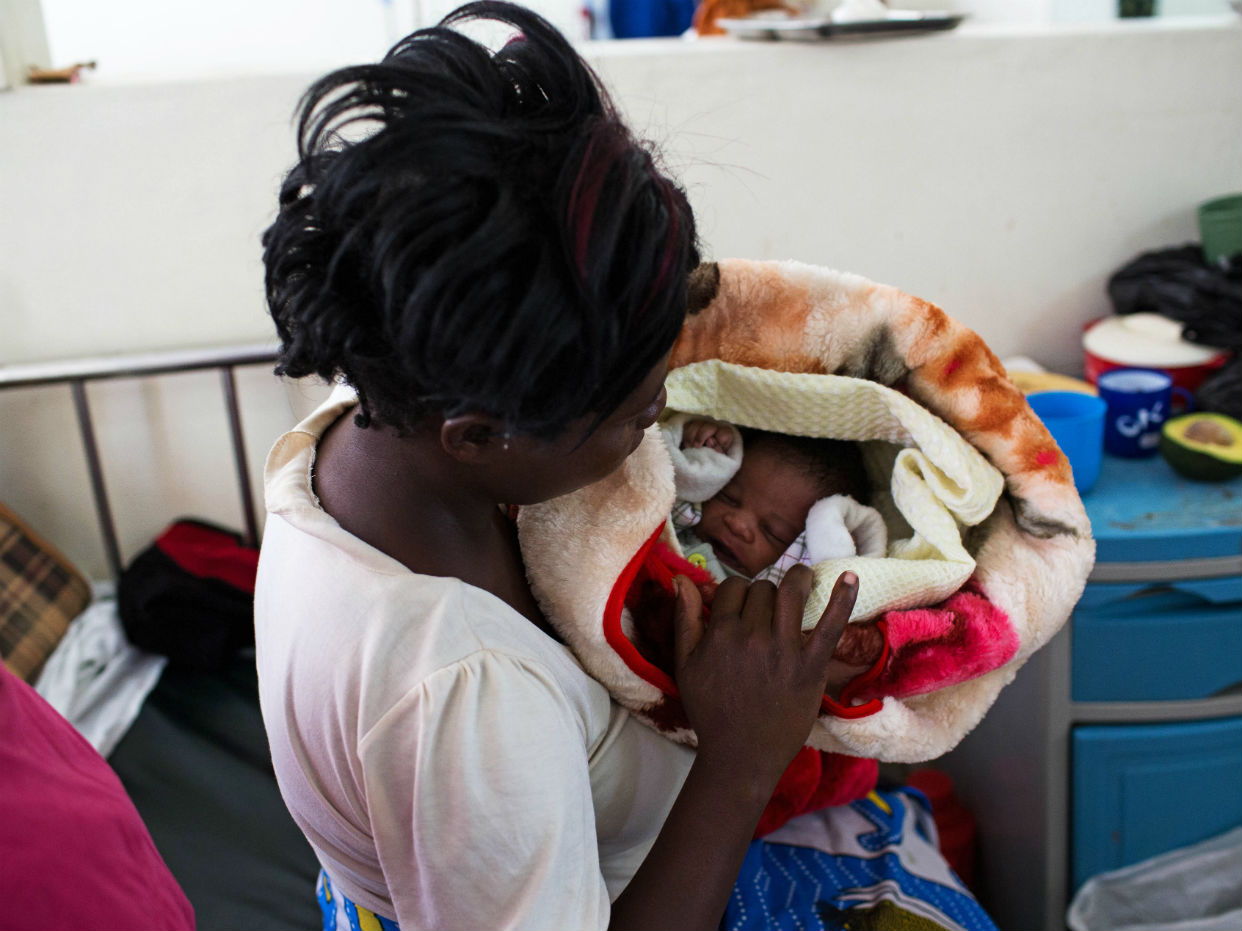
Children in Ghana, Kenya and Malawi have been selected to trial the world's first malaria vaccine, which the World Health Organisation (WHO) says could save "tens of thousands of lives".
Around 750,000 children aged between five months and 17 months will receive injections designed to reduce their risk of contracting malaria, which kills more than 400,000 people per year.
How does the vaccine work?
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
British pharmaceutical giant GlaxoSmithKline's RTS,S, also known by its trade name Mosquirix, "trains the immune system to attack the malaria parasite, which is spread by mosquito bites", says the BBC.
Trials showed it cut the risk of malaria by 40 per cent. WHO regional director Dr Matshidiso Moeti said that, combined with existing measures such as mosquito nets and insecticides, the vaccine could save "tens of thousands" of lives.
Why only three countries?
Ghana, Kenya and Malawi were chosen "because all have strong prevention and vaccination programs but continue to have high numbers of malaria cases".
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
To be effective, the vaccine has to be administered once a month for three months, then again 18 months later, which could be challenging in poor and remote regions that only have sporadic access to healthcare.
The three pilot schemes will determine whether larger-scale implementation is feasible.
"Information gathered in the pilot programme will help us make decisions on the wider use of this vaccine," Moeti said.
