The FDA just approved a high-tech tiara to prevent migraines, without drugs
YouTube

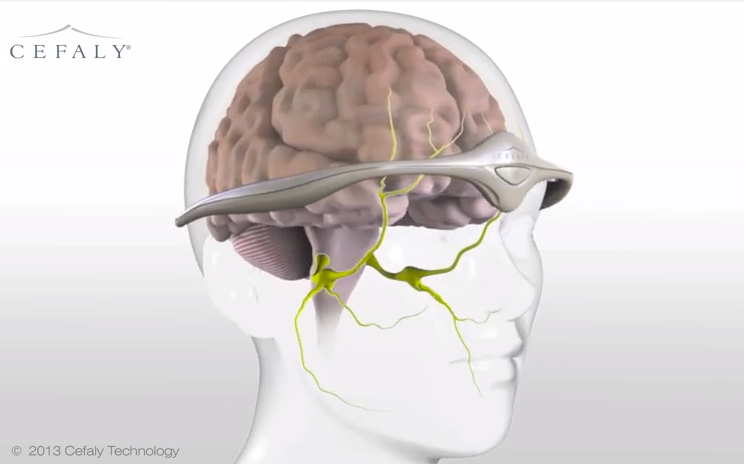
In potentially great news for migraine sufferers, the Food and Drug Administration has approved a plastic headband-like device that purports to not just treat but also prevent migraines, without using any medication. Instead of drugs, the "transcutaneous electrical nerve stimulation" (TENS) headband delivers a small electric current through an electrode on the forehead. The electricity stimulates the trigeminal nerve, which is tied to migraines. Belgium's STX-Med makes the battery-powered tiara, called Cefaly, which is now available in the U.S. with a prescription. STX-Med describes how it works in greater detail in the video below. --Peter Weber
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
Peter has worked as a news and culture writer and editor at The Week since the site's launch in 2008. He covers politics, world affairs, religion and cultural currents. His journalism career began as a copy editor at a financial newswire and has included editorial positions at The New York Times Magazine, Facts on File, and Oregon State University.
