Solving COVID: April 28, 2021
Israel's falling death rate, Pfizer's COVID-19 pill, and more

- 1. Where things stand
- 2. CDC says fully vaccinated people generally don't need masks outdoors
- 3. Biden administration sharing AstraZeneca coronavirus vaccine abroad
- 4. Israel recorded zero COVID-19 deaths in a day for the first time in 10 months
- 5. Pfizer CEO says oral COVID-19 drug could be ready by end of year
1. Where things stand
The United States has vaccinated nearly 142 million people — roughly 53 percent of the population over age 16. Encouragingly, the so-called "fourth wave" that began in early April seems to be subsiding, with average new daily cases falling more than 20 percent over the last two weeks. The pace of vaccinations has slowed, however, from a high of more than 4 million doses administered per day in early April to just 1.6 million doses administered on April 27. Some mass vaccination sites are closing up shop as vaccine supply outstrips demand and the national focus turns to encouraging more hesitant Americans to get the jab. According to The New York Times, "White House and health officials are comparing the next phase of the vaccination campaign to a get-out-the-vote effort." Some states are getting creative: West Virginia, for example, will give young people a $100 savings bond if they get vaccinated. Global cases remain high, and this week Dr. Anthony Fauci called for a more global response, saying "a global response means equity throughout the world."
The Washington Post The New York Times
2. CDC says fully vaccinated people generally don't need masks outdoors
The Centers for Disease Control and Prevention updated its guidelines on Tuesday to say that it's generally safe for those who have been fully vaccinated against COVID-19 to go maskless outdoors when not in crowded settings. A CDC graphic outlined various situations in which fully vaccinated people don't need to wear a mask, including walking, running, or biking outdoors with members of their household or attending small outdoor gatherings. Fully vaccinated people can also safely dine outdoors with friends from multiple households without a mask. The guidance said unvaccinated people also don't need to wear a mask while walking, running, or biking outdoors with household members and while attending small, outdoor gatherings with fully vaccinated family and friends. But it still recommended unvaccinated people wear a mask at outdoor gatherings with other unvaccinated people.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
3. Biden administration sharing AstraZeneca coronavirus vaccine abroad
The United States will send other countries its supply of the COVID-19 vaccine developed by AstraZeneca and the University of Oxford, the White House said Monday. The Oxford-AstraZeneca vaccine has not been granted emergency use authorization by the Food and Drug Administration yet, and White House COVID-19 coordinator Jeff Zients said the country doesn't need the supply given "the strong portfolio of vaccines" already available in the U.S. The Biden administration previously shared about 4 million doses of the Oxford-AstraZeneca vaccine with neighboring Canada and Mexico. Now the Biden administration plans to send doses to other countries desperate to step up their vaccination campaigns. About 10 million doses have been produced and await FDA quality inspection. Another 50 million doses should be ready by June.
The Associated Press The Washington Post
4. Israel recorded zero COVID-19 deaths in a day for the first time in 10 months
Israel recorded no new daily COVID-19 deaths on Friday for the first time in 10 months. At that point, the country had enacted strict lockdown measures to curb the spread of the coronavirus, but that's not the case now. Cases are plummeting even as Israel has gradually been lifting restrictions, including an outdoor mask mandate. So far, the evidence strongly suggests that the decline is largely thanks to a swift and successful vaccine rollout. Israel has the highest vaccination rate in the world: About 56 percent of the population has received two doses, which is required for the Pfizer-BioNTech vaccine that's been in use in Israel.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
5. Pfizer CEO says oral COVID-19 drug could be ready by end of year
Pfizer CEO Albert Bourla said Tuesday that the company's experimental oral medicine for COVID-19 could be available by the end of 2021. "If all goes well, and we implement the same speed that we are, and if regulators do the same, and they are, I hope that by the end of the year," Bourla told CNBC's Squawk Box. The company began early-stage clinical trials for its new antiviral coronavirus therapy in March. The drug is a protease inhibitor designed to target an enzyme the virus uses to replicate in human cells. Pfizer partnered with German drugmaker BioNTech to make the first coronavirus vaccine to be approved for emergency use in the United States. It also has requested FDA approval to authorize use of its vaccine on adolescents ages 12 to 15, and it is testing the vaccine on children 6 months to 11 years old.
-
 Art review: Diane Arbus: Constellation
Art review: Diane Arbus: ConstellationFeature Park Avenue Armory, New York City, through Aug. 17
-
 July fiction: Summers to remember
July fiction: Summers to rememberFeature Featuring the latest summer-themed novels from Darrow Farr, Lucas Schaefer, and more
-
 Why are flash floods in Texas so deadly?
Why are flash floods in Texas so deadly?Today's Big Question Over 100 people, including 27 girls at a summer camp, died in recent flooding
-
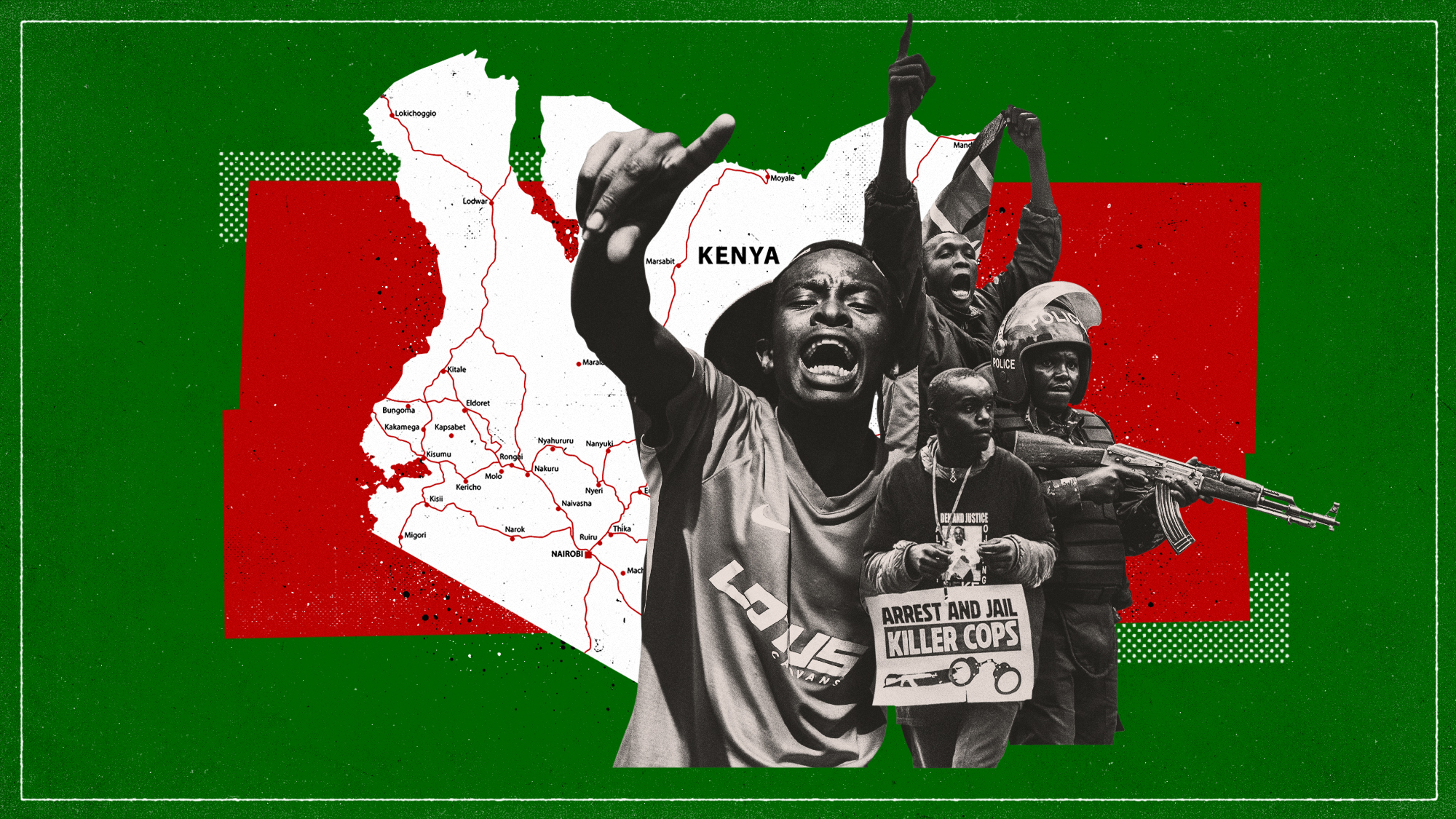 One year after mass protests, why are Kenyans taking to the streets again?
One year after mass protests, why are Kenyans taking to the streets again?today's big question More than 60 protesters died during demonstrations in 2024
-
 What happens if tensions between India and Pakistan boil over?
What happens if tensions between India and Pakistan boil over?TODAY'S BIG QUESTION As the two nuclear-armed neighbors rattle their sabers in the wake of a terrorist attack on the contested Kashmir region, experts worry that the worst might be yet to come
-
 Why Russia removed the Taliban's terrorist designation
Why Russia removed the Taliban's terrorist designationThe Explainer Russia had designated the Taliban as a terrorist group over 20 years ago
-
 Inside the Israel-Turkey geopolitical dance across Syria
Inside the Israel-Turkey geopolitical dance across SyriaTHE EXPLAINER As Syria struggles in the wake of the Assad regime's collapse, its neighbors are carefully coordinating to avoid potential military confrontations
-
 'Like a sound from hell': Serbia and sonic weapons
'Like a sound from hell': Serbia and sonic weaponsThe Explainer Half a million people sign petition alleging Serbian police used an illegal 'sound cannon' to disrupt anti-government protests
-
 The arrest of the Philippines' former president leaves the country's drug war in disarray
The arrest of the Philippines' former president leaves the country's drug war in disarrayIn the Spotlight Rodrigo Duterte was arrested by the ICC earlier this month
-
 Ukrainian election: who could replace Zelenskyy?
Ukrainian election: who could replace Zelenskyy?The Explainer Donald Trump's 'dictator' jibe raises pressure on Ukraine to the polls while the country is under martial law
-
 Why Serbian protesters set off smoke bombs in parliament
Why Serbian protesters set off smoke bombs in parliamentTHE EXPLAINER Ongoing anti-corruption protests erupted into full view this week as Serbian protesters threw the country's legislature into chaos