Solving COVID: March 3, 2021
America gets a third vaccine, Biden promises enough doses for every U.S. adult by May, and more

- 1. FDA grants emergency use authorization for Johnson & Johnson COVID-19 vaccine
- 2. Trial data suggest J&J's vaccine stands up well against worrying variants
- 3. Merck to help make Johnson & Johnson's COVID-19 vaccine
- 4. Biden promises enough vaccine for every U.S. adult by May
- 5. Moderna to start trials of booster shot for COVID-19 variant
1. FDA grants emergency use authorization for Johnson & Johnson COVID-19 vaccine
The Food and Drug Administration on Saturday authorized Johnson & Johnson's single-dose COVID-19 vaccine for emergency use, which means there are now three effective shots available for Americans. The Johnson & Johnson candidate registered a 72 percent efficacy rate in the U.S. clinical trial. That falls short of the roughly 95 percent rates seen in the Pfizer-BioNTech and Moderna trials, but experts are still very pleased with J&J's results, especially since it also showed 85 percent efficacy against severe COVID-19 infections, and 100 percent efficacy against hospitalizations and deaths, suggesting it will be a crucial tool in the fight to end the pandemic.
2. Trial data suggest J&J's vaccine stands up well against worrying variants
One very encouraging datapoint in the FDA's analysis of the single-dose Johnson & Johnson COVID-19 vaccine suggests the shot works in areas where highly contagious variants are spreading, like Brazil and South Africa. The overall efficacy rate — that is, protection against any symptomatic infection — in the South African trial was lower than it was in the United States initially, but the numbers started to even out over time. After a month, the shot's efficacy rate against severe infection was 82 percent. The figures out of Brazil show a similar trajectory, though the efficacy rate against severe infections was actually slightly higher than in the U.S. The trial data is not a guarantee of the vaccine's effectiveness in a real-world setting, but the FDA's breakdown should help alleviate growing concerns that the variants can completely resist vaccinations.
Subscribe to The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
3. Merck to help make Johnson & Johnson's COVID-19 vaccine
President Biden's administration unveiled a major partnership between Merck & Co. and Johnson & Johnson to manufacture the latter's COVID-19 vaccine. Merck will help make the single-shot Johnson & Johnson vaccine, a move that "could sharply boost the supply," The Washington Post reports. Merck will reportedly dedicate two U.S. facilities to the effort, though it could take months to get these facilities running. The Biden administration sought a partnership after it became clear that Johnson & Johnson was behind on vaccine production. The company has said it expects to have 20 million vaccine doses available by the end of March and 100 million by the end of June. Merck previously attempted to develop COVID-19 vaccines but abandoned the effort due to inadequate immune responses.
4. Biden promises enough vaccine for every U.S. adult by May
President Biden said Tuesday that the "stepped-up process" for producing and distributing coronavirus vaccines had put the country "on track" to have enough doses for every adult in the United States "by the end of May," several months earlier than he previously predicted. Biden said the federal government was giving Johnson & Johnson, maker of a vaccine just approved for emergency use, the support it needs for around-the-clock production. The Biden administration brokered a deal under which Merck agreed to produce Johnson & Johnson's single-dose COVID-19 vaccine to boost the nation's inoculation effort. Johnson & Johnson's vaccine gives the U.S. a third vaccine, along with those produced by Pfizer and Moderna. A Merck spokesperson said the drug maker "remains steadfast in our commitment to contribute to the global response to the pandemic and to preparing to address future pandemics."
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
The New York Times The Wall Street Journal
5. Moderna to start trials of booster shot for COVID-19 variant
Moderna announced last week it will begin testing a new version of its COVID-19 vaccine designed to target the coronavirus variant first reported in South Africa. The pharmaceutical company said it has sent doses of the booster shot to the U.S. National Institutes of Health for clinical trials. In a statement, CEO Stéphane Bancel said Moderna is "committed to making as many updates to our vaccine as necessary until the pandemic is under control." Moderna previously revealed that preliminary studies showed the vaccine still made neutralizing antibodies above protective levels for the South African variant, but because it was a reduced level, it prompted the company to begin tweaking the vaccine against the strain, as well as variants that first spread in the United Kingdom and Brazil.
-
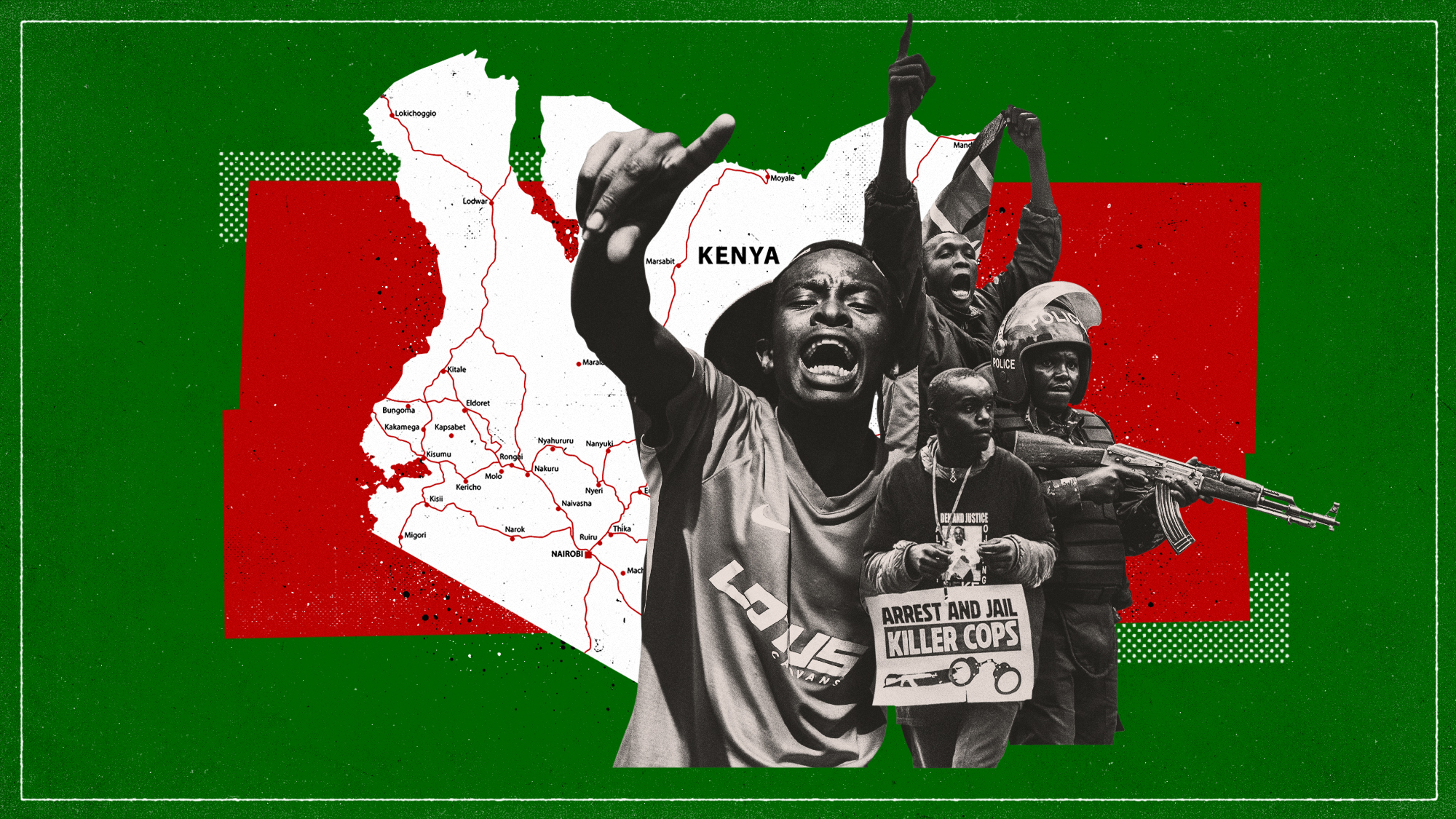 One year after mass protests, why are Kenyans taking to the streets again?
One year after mass protests, why are Kenyans taking to the streets again?today's big question More than 60 protesters died during demonstrations in 2024
-
 What happens if tensions between India and Pakistan boil over?
What happens if tensions between India and Pakistan boil over?TODAY'S BIG QUESTION As the two nuclear-armed neighbors rattle their sabers in the wake of a terrorist attack on the contested Kashmir region, experts worry that the worst might be yet to come
-
 Why Russia removed the Taliban's terrorist designation
Why Russia removed the Taliban's terrorist designationThe Explainer Russia had designated the Taliban as a terrorist group over 20 years ago
-
 Inside the Israel-Turkey geopolitical dance across Syria
Inside the Israel-Turkey geopolitical dance across SyriaTHE EXPLAINER As Syria struggles in the wake of the Assad regime's collapse, its neighbors are carefully coordinating to avoid potential military confrontations
-
 'Like a sound from hell': Serbia and sonic weapons
'Like a sound from hell': Serbia and sonic weaponsThe Explainer Half a million people sign petition alleging Serbian police used an illegal 'sound cannon' to disrupt anti-government protests
-
 The arrest of the Philippines' former president leaves the country's drug war in disarray
The arrest of the Philippines' former president leaves the country's drug war in disarrayIn the Spotlight Rodrigo Duterte was arrested by the ICC earlier this month
-
 Ukrainian election: who could replace Zelenskyy?
Ukrainian election: who could replace Zelenskyy?The Explainer Donald Trump's 'dictator' jibe raises pressure on Ukraine to the polls while the country is under martial law
-
 Why Serbian protesters set off smoke bombs in parliament
Why Serbian protesters set off smoke bombs in parliamentTHE EXPLAINER Ongoing anti-corruption protests erupted into full view this week as Serbian protesters threw the country's legislature into chaos


