The Justice Department wants to stop the FDA from regulating death-penalty drugs

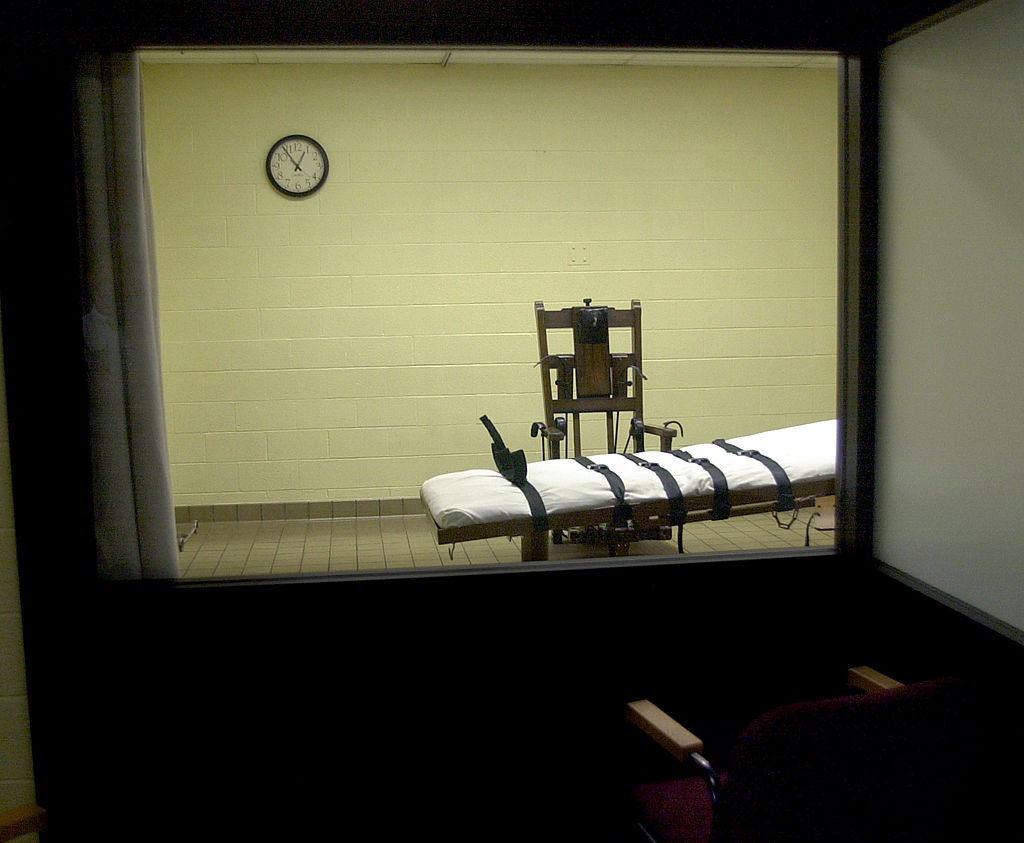
In an opinion earlier this month, the Justice Department's Office of Legal Counsel decided that the Food and Drug Administration "lacks jurisdiction" over drugs used to kill inmates through lethal injection, The Washington Post reports. The Justice Department is siding with Texas, which sued the FDA in early 2017 over the agency's 2015 seizure of 1,000 vials of the anesthetic sodium thiopental — once commonly used in lethal-injection cocktails — from an unregistered overseas distributor.
The issue has caused tension in the Trump administration. More than a year ago, the Post reports, Attorney General Jeff Sessions and FDA Commissioner Scott Gottlieb — both of whom left the administration last year — had a heated argument in the White House Situation Room, with Sessions demanding that Gottlieb allow execution drugs into the U.S. without any scrutiny and Gottlieb refusing.
The Justice Department's new opinion is pretty sweeping, arguing that "articles intended for use in capital punishment by a state or the federal government cannot be regulated as 'drugs' or 'devices'" by the FDA. But the opinion applies only to the death penalty, the OLC added, not whether the FDA "has jurisdiction over drugs intended for use in physician-assisted suicide."
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
It isn't clear what affect the OLC decision will have. Imports of sodium thiopental have been blocked under a federal injunction since 2012. Hospira, the sole U.S. maker of sodium thiopental, stopped producing it in 2011, citing its use in capital punishment. The OLC opinion seems aimed at "giving a green light" to states to import execution drugs from China, India, and other countries that don't object to their use in executions, Deborah Denno, a law professor at Fordham University, tells the Post. "It has the potential to open the floodgates."
But the 2012 injunction is still in effect for now, the Post notes, and "it is not clear whether the Justice Department will seek to have that injunction lifted, a move that could spark a long legal tussle."
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Peter has worked as a news and culture writer and editor at The Week since the site's launch in 2008. He covers politics, world affairs, religion and cultural currents. His journalism career began as a copy editor at a financial newswire and has included editorial positions at The New York Times Magazine, Facts on File, and Oregon State University.

