FDA to re-evaluate effectiveness of common nasal congestion ingredient

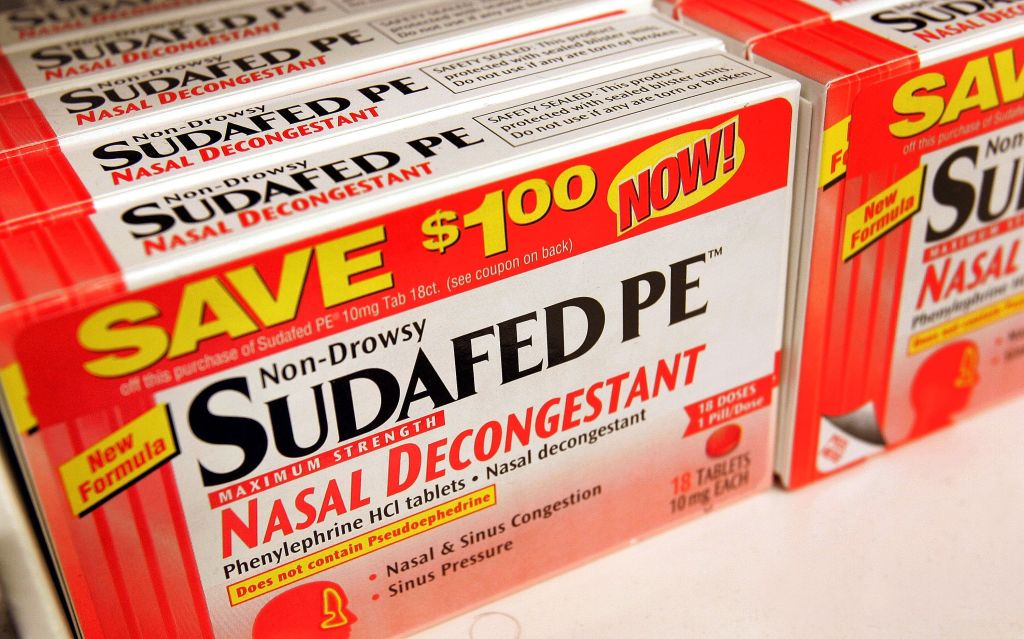
The Food and Drug Administration (FDA) will convene a panel next week to re-evaluate the effectiveness of oral phenylephrine, a common ingredient found in numerous over-the-counter decongestants. The panel is slated meet Monday and Tuesday, just days after the FDA released a report claiming that phenylephrine likely doesn't work.
The panel will examine the potential benefits and drawbacks of phenylephrine and question medical advisers. The drug, which was first approved by the FDA in the 1970s, can be found in many varieties of anti-cold medications, including versions of Nyquil, Sudafed, Benadryl, Vicks and Mucinex.
These medications are currently classified as being "generally recognized as safe and effective," according to an FDA fact sheet. However, the FDA's recent report, released last Thursday, claimed that phenylephrine is unlikely to work in any dosages, putting that classification at high risk of being revoked.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
"Because this would represent a major change in the agency's position, we believe that presenting this information in an open public forum, along with a full discussion and vote" from committee members, "will be extremely helpful," the FDA's report said.
If phenylephrine does end up being reclassified, major drug companies like Kenvue, Procter & Gamble, Reckitt Benckiser and others would likely have to reformulate many of their flagship decongestants or pull them from store shelves, Bloomberg reported.
The effectiveness of phenylephrine has long been debated. A 2015 clinical trial of 500 adults with seasonal allergies found that phenylephrine was "not significantly better than placebo at relieving nasal congestion in adults." Dr. Wynne Armand, a primary care doctor at Massachusetts General Hospital, told NBC News she advises patients with cold symptoms "to avoid buying oral meds that have phenylephrine," and won't prescribe it.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Justin Klawans has worked as a staff writer at The Week since 2022. He began his career covering local news before joining Newsweek as a breaking news reporter, where he wrote about politics, national and global affairs, business, crime, sports, film, television and other news. Justin has also freelanced for outlets including Collider and United Press International.

