Can malaria be wiped out?
Malawi begins roll-out of landmark vaccine for children
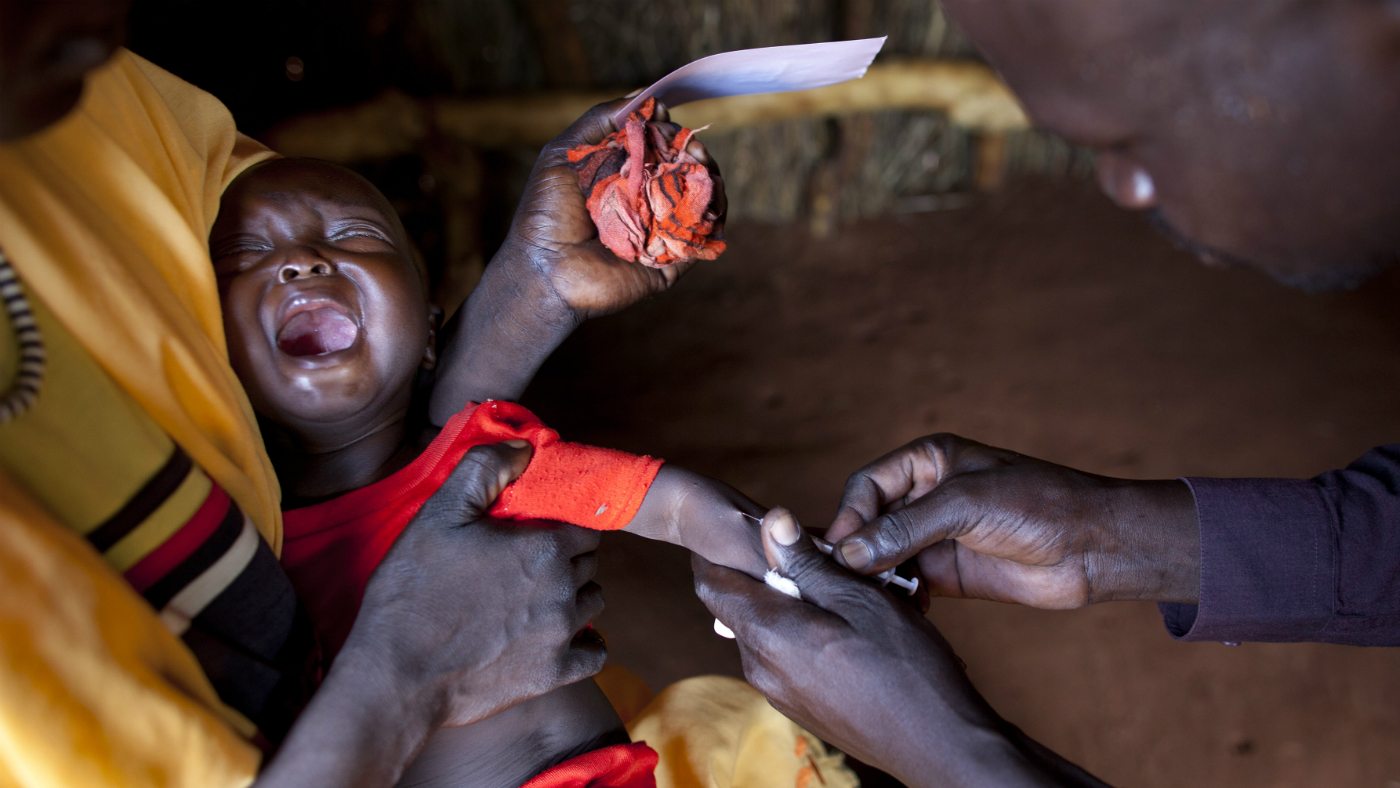
The southeast African nation of Malawi is rolling out a pioneering new treatment that is said to be the first to give partial protection against malaria.
The BBC reports that the vaccine, known as RTS,S, trains the immune system to attack the malaria parasite and has been “more than three decades in the making”.
Following stringent testing - and investments from NGOs totalling an estimated $1bn (£770m) - the landmark trial will be rolled out this week in Malawi’s capital, Lilongwe, and then in Kenya and Ghana next week.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
The World Health Organization (WHO) has issued a statement welcoming the trials, during which a total of around 120,000 children aged two years and under will be immunised.
“We have seen tremendous gains from bed nets and other measures to control malaria in the last 15 years, but progress has stalled and even reversed in some areas,” said Tedros Adhanom Ghebreyesus, WHO director-general. “The malaria vaccine has the potential to save tens of thousands of children’s lives.”
Previous, much smaller trials found that nearly 40% of children aged between five and 17 months who received RTS,S were protected from malaria.
So are we on the verge of eradicating malaria?
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
What is malaria?
Malaria is a tropical disease of the blood that is caused by the Plasmodium parasite, which is transmitted from person to person by a bite from the female Anopheles mosquito.
The US Center for Disease Control (CDC) reports that symptoms of less severe malaria include “fever and flu-like illness, including shaking chills, headache, muscle aches, and tiredness”, while nausea, vomiting, and diarrhoea may also occur.
Severe malaria can cause anemia and jaundice, and if not promptly treated, kidney failure, seizures, mental confusion, coma and ultimately death.
Deutsche Welle reports that the RTS,S vaccine trials follow “a resurgence in malaria case globally”, with 219 million people diagnosed in 2017, an increase of two million on the previous year.
Around 435,000 people die from the disease each year around the world, “mostly in Africa and most of them children under the age of five”, the German newspaper adds.
How effective is the vaccine?
Also known by the trade name Mosquirix, the protein-based vaccine was developed by GlaxoSmithKline and has already undergone five years of clinical trials on 15,000 people in seven countries, Al Jazeera reports.
According to the WHO website, “overall, there were 29% fewer cases of severe malaria in children who received the vaccine”.
Scientists say if rolled out on a large scale, the vaccine may surpass mosquito nets, insecticides and drugs as a key tool in combating the disease.
Matshidiso Moeti, WHO regional director for Africa, said that the launch of the new trials marks “a day to celebrate” as humans “change the trajectory of malaria through childhood vaccination”.
Will it eradicate malaria?
The Japan Times says that although RTS,S is so far the most effective treatment of its type against malaria, it “will not give full protection against the mosquito-borne disease” and is not sufficient to eradicate the disease entirely.
The sheer scale of the task of eradication is evident in the numbers. In recorded history, only one human disease has been eradicated from existence - smallpox.
Eradicating malaria may only be possible by using an extremely controversial gene-drive technology known as CRISPR.
Described by Vox as “humanity’s single best chance to eradicate malaria”, CRISPR is a relatively new technology that has made it easier to edit the genetic make-up of a species by changing the DNA and quickly propagating a chosen gene throughout a population.
Two possible methods to wipe out the disease have been developed already. In 2016, a team of scientists at the University of California announced that they had developed a gene that makes the Anopholes mosquito immune to the malaria parasite and that could be introduced into the wider mosquito population using CRISPR.
And last year, scientists at Imperial College London used the technology to effectively spread an edited gene that makes the female Anopheles mosquito that transmits malaria completely infertile.
Nature journal says that this technique “has the potential to eradicate disease, control pests and alter entire ecosystems”, but may also have “unpredictable consequences”.
Washington DC-based news site NPR adds that the move could “inadvertently have a negative effect on crops, for example, by eliminating important pollinators”, while a population crash could also pave the way for an increase in other mosquitoes carrying other diseases.
“We can't be taking lightly this extermination technology,” says Dana Perls of anti-GMO organisation Friends of the Earth. “We need to hit the pause button on gene drives.”
