Eye drops recalled after being linked to drug-resistant infections

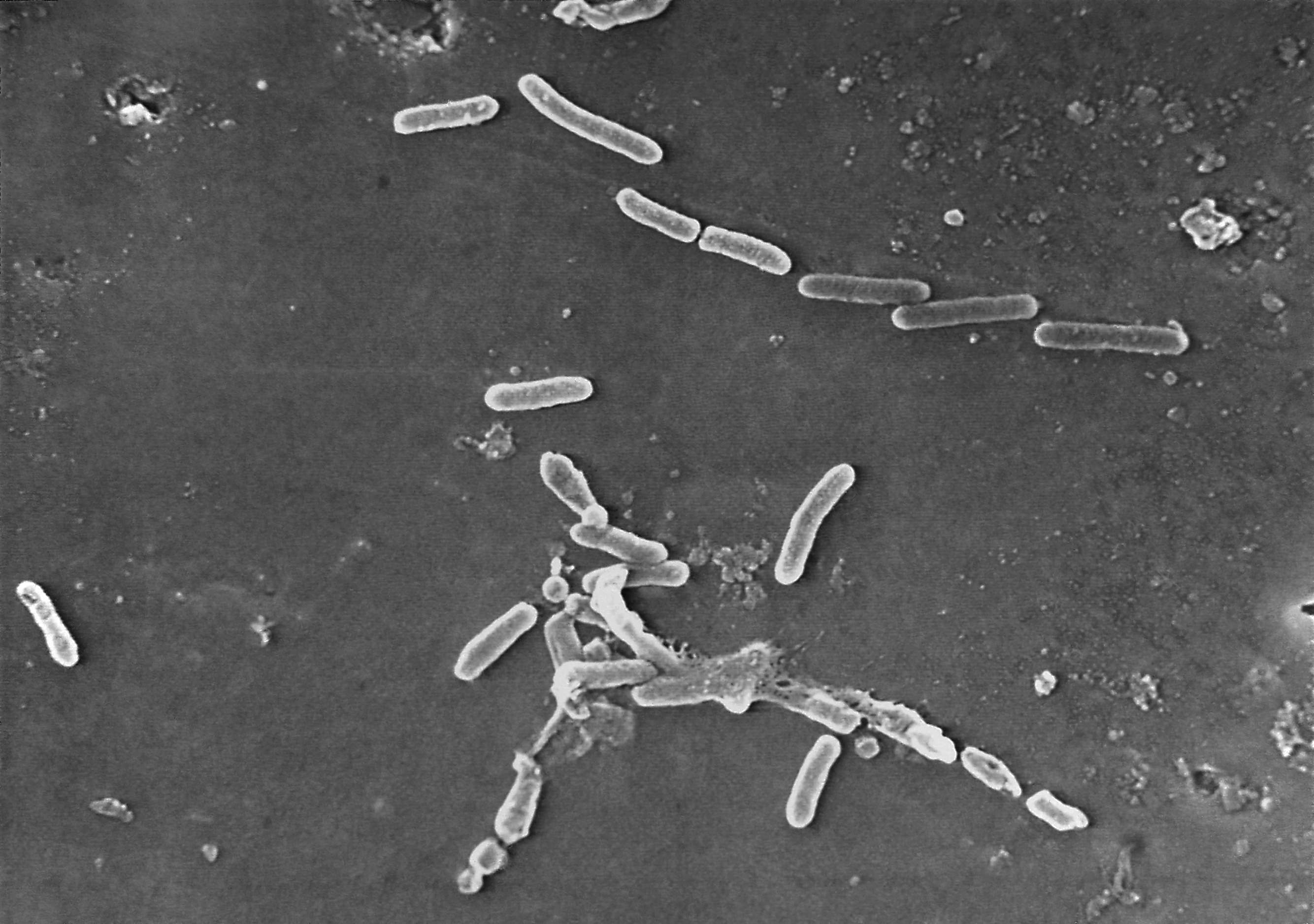
India's Global Pharma, the manufacturer of EzriCare Artificial Tears, is recalling the product in the United States, after it was linked to an outbreak of drug-resistant infections in 12 states.
At least 55 people have reported infections, including of the blood and lungs, caused by the Pseudomonas aeruginosa bacteria, The Associated Press reports, with one person dying and five experiencing permanent vision loss. Cases have been reported in California, Colorado, Connecticut, Florida, New Jersey, New Mexico, New York, Nevada, Texas, Utah, Washington, and Wisconsin.
EzriCare Artificial Tears was available over the counter, and used as a lubricant to treat eye irritation and dryness. Global Pharma said so far, it is unaware of any evidence that definitively links EzriCare to the infections, and the Food and Drug Administration said it recommended recalling the product due to a lack of testing and proper controls on packaging.
The Week
Escape your echo chamber. Get the facts behind the news, plus analysis from multiple perspectives.

Sign up for The Week's Free Newsletters
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
From our morning news briefing to a weekly Good News Newsletter, get the best of The Week delivered directly to your inbox.
A free daily email with the biggest news stories of the day – and the best features from TheWeek.com
Catherine Garcia has worked as a senior writer at The Week since 2014. Her writing and reporting have appeared in Entertainment Weekly, The New York Times, Wirecutter, NBC News and "The Book of Jezebel," among others. She's a graduate of the University of Redlands and the Columbia University Graduate School of Journalism.

